By Mounir Raji, Senior Scientist
Isotope-labeled compounds have emerged as important tools in drug discovery and development. The incorporated isotopes, particularly deuterium, in pharmaceuticals, have been shown to improve ADME properties compared to their non-isotopically labeled counterparts. Since 2006, the FDA has approved >10 drugs labeled with stable isotopes such as deutetrabenazine (deuterated), deucravacitinib (deuterated) and lubiprostone (C13-labeled).
Traditionally, isotope-labeled compounds are generated by different methods, e.g., organic chemistry routes in batches using isotopically enriched starting materials. At X-Chem, we took two synthetic directions to improve the cost-effectiveness, regioselectivity, versatility and the yield of isotope-labeling reactions. We specifically developed two methodologies to address current challenges in deuterium- and C13-labeling:
- Using late-stage direct hydrogen isotopic exchange (HIE) instead of isotopically enriched starting materials: Direct isotopic exchange allows us to perform the labeling without the need for prefunctionalization. It’s also cost-efficient, due to the availability of reagents, such as D2O or 13CO2. Atom-economy-wise, the labeling can be introduced at a specific position without constructing the entire molecule, which adds to another advantage — more applicable to synthesizing complex natural products or pharmaceuticals.
- Using flow synthesis instead of batch synthesis: Flow chemistry can offer numerous advantages compared to traditional batch chemistry, such as precise control of reactions parameters (e.g., temperature, reaction time), improved mixing and enhanced safety. We surmised that isotope labeling can also benefit from flow conditions.
Highly selective deuterium labeling
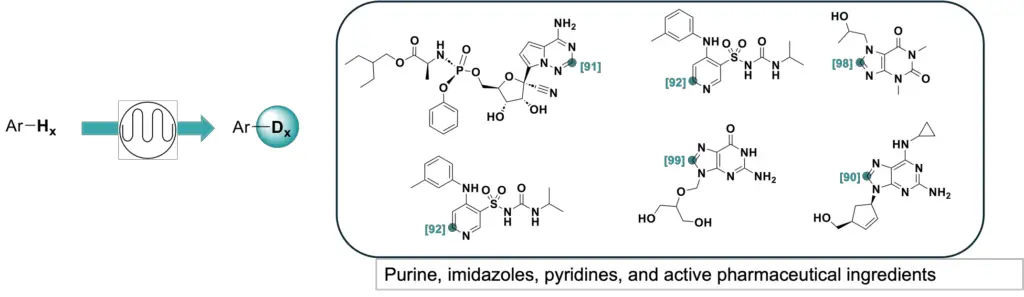
Inspired by Sajiki’s work using Pd/C, D2O and catalytic amount of D2 gas to label nucleobases, we screened different substrates with several readily available heterogenous catalysts under flow conditions. Of all the tests, Raney nickel had shown superior deuterium incorporation and could be fine-tuned by pretreatment with caffeine. From those results, we then optimized the reactions to develop a continuous flow Raney nickel-catalyzed HIE process, which was published in Advanced Synthesis & Catalysis. The process is compatible with nitrogen-containing heterocycles and pharmaceutical compounds, as we successfully labeled various purine bases, imidazole, pyridine as well as complex pharmaceuticals such as abacavir and remdesivir at high yields.
Controlled ¹³CO2 gas generation
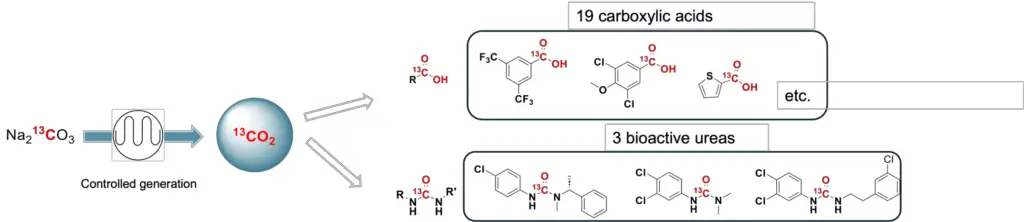
The most common source of ¹³C is ¹³CO₂ gas, which is typically available in cylinders. Achieving sufficient mixing and controlling the stoichiometry of multiphase (e.g., gas-liquid) reactions can be challenging. Moreover, these cylinders often necessitate excess pressure and are costly.
As a solution, we developed an on-demand ¹³CO₂ flow chamber.
The chamber is based on a “tube-in-tube” flow reactor. This gas-liquid reactor comprises of a Teflon membrane encased in a PTFE tube. This design leverages the principle of diffusion to facilitate the transfer of the reactive gas into the solution stream during the synthesis. The use of Na213CO3 as a precursor allows for greater control over reaction stoichiometry, yielding a series of C13-labeled products such as bioactive ureas and 13C-carboxylic acids with high efficiency.
Applications Beyond Pharmaceuticals
Deuterated and C13-labeled compounds are used in various fields even beyond pharmaceuticals or metabolic studies. For example, in mass spectrometry, labeled compounds serve as internal standards for quantification. In kinetic isotope effect studies, labeled compounds are used to interrogate reaction mechanisms and transition state structures. We envision that with our improved synthetic methods, isotopically labeled compounds could be generated more efficiently and cost-effectively.


