X‑Chem’s DNA-Encoded Library (DEL) Platform Enables the Discovery of Lead-Like Molecules for Medicinal Chemistry Progression
Discovery of Novel, Potent Inhibitors of Hydroxy Acid Oxidase 1 (HAO1) Using DNA-Encoded Chemical Library Screening
X-Chem’s DNA-encoded library (DEL) technology platform was created to feed lead-like hits into traditional medicinal chemistry. We recently published the discovery of several inhibitors of hydroxy acid oxidase 1 (HAO1), which exemplifies our integrated approach, in the Journal of Medicinal Chemistry. The result produced several high-quality leads, representing a unique chemical class relative to previous inhibitors.
HAO1 as a validated drug target
Primary hyperoxaluria type 1 (PH1), an inherited rare liver disorder of glyoxylate metabolism, arises from mutations in the enzyme alanine-glyoxylate aminotransferase (AGXT). AGXT converts glyoxylate, the product of hydroxy acid oxidase 1 (HAO1), to glycine in the liver. Defective glyoxylate metabolism in PH1 patients leads to the accumulation of toxic levels of oxalate, which results in progressive kidney deterioration, and ultimately, end-stage renal disease. Inhibition of hydroxy acid oxidase 1 (HAO1) is a strategy used to mitigate the accumulation of toxic oxalate that is caused by the reduced activity of AGXT in PH1 patients. Currently, there are no approved treatments for PH1, leaving patients in need of both liver and kidney transplants. Although siRNA therapy targeting HAO1 in clinical trials has shown promising outcomes and has validated this therapeutic strategy, small-molecule drug discovery efforts have been limited to optimizing CCPST or CDST series, known HAO1 inhibitors of low to modest potencies. X-Chem’s DEL screening platform is well-suited for the discovery of novel and potent enzyme inhibitors. With a paucity of existing inhibitors and an unaddressed medical need, HAO1 was an ideal target for our powerful discovery platform.
From functionally active library compounds to X-ray crystallography
Distinct hit series emerged from the affinity-mediated selection of X-Chem’s collection of DEL libraries against human HAO1 (hHAO1) protein. Compounds 5 and 6, derived from our proprietary oxadiazole library, were of high interest due to their high ligand efficiency (LE of 0.36), and superb physicochemical properties. In addition, these library compounds showed dose-dependent activity in a cell-based phenotypic rescue assay, a stringent assay indicating the inhibitory effect of the small molecule series against hHAO1 in CHO-GO cells.
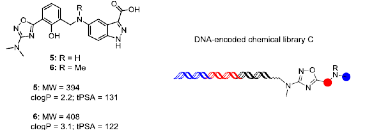

Adapted with permission from J. Med. Chem. 2021, 64, 10, 6730–6744, Copyright © 2021 American Chemical Society
We initiated a medicinal chemistry campaign focused on improving the permeability of these series while maintaining metabolic stability, applying a general tactic of lowering the tPSA and increasing the lipophilicity. We optimized the amino oxadiazole region, a constant region where the small molecules link to the DNA tags in the original context of the DNA-encoded library, which resulted in compound 29. Compound 29 exhibited improved permeability, exposure and oral bioavailability, with sufficient plasma exposure for in vivo efficacy assessment.
It was exciting to see X-ray co-crystal structures of the initial DEL hit series in complex with hHAO1.

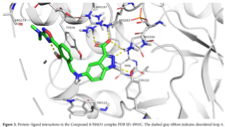
Reprinted with permission from J. Med. Chem. 2021, 64, 10, 6730–6744, Copyright © 2021 American Chemical Society
The X-ray crystal structure of compound 5 further enabled structure-based drug design (SBDD), which we used to generate non-carboxylic acid containing HAO1 inhibitors. Traditional docking studies of analogues bearing isosteric acid replacement of the indazole acid in compound 5 led to the confirmation of the non-acid HAO1 inhibitor, compound 31, which is an appealing starting point for future optimization.
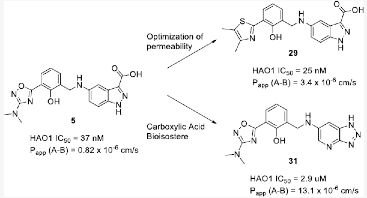
Reprinted with permission from J. Med. Chem. 2021, 64, 10, 6730–6744, Copyright © 2021 American Chemical Society
X-Chem’s advantages for drug discovery lead optimization
The discovery of multiple inhibitors of HAO1 highlights the value of X-Chem’s integrated approach. By biasing our libraries toward molecules exhibiting lead-like properties, we give our clients the best chance of a lead-like hit. With a high-quality starting point, we can use familiar medchem methodologies to reach better optimized leads.
This successful HAO1 inhibitor search demonstrates how we integrate DEL with conventional approaches to drive our clients toward their drug discovery goals. For a more complete account of this work, including a description of additional hit compounds, please see our paper in Journal of Medicinal Chemistry.
Log KIAM Is the New LogD
By Johan Bartholomeus, Senior Principal Scientist, and Philippe McGee, Principal Scientist In a drug discovery project, during the hit-to-lead and...
X‑Chem and the SGC are pioneering crowd-sourced AI advancements by making DEL screening data public.
Artificial intelligence (AI) is now recognized as an indispensable component of the modern drug discovery tool kit, applied at all...